- Home Page
- Company Profile
-
Our Products
- Pharmaceutical Tablets
- Sodium Bicarbonate Tablets
- Ferrous Sulphate and Folic Acid
- Prochlorperazine Tablets
- Fexofenadine HCL Tablets
- Ciprofloxacin Tablets 500mg
- Sulfadiazine Tablet
- Cefuroxime Axetile Tablets
- Artemether and Lumefantrine Tablets
- Nifedipine Extended Release Tablets USP 20 mg
- Vitamin D3 Tablets
- Quinine Sulphate Tablet 300 mg
- Clomifene Tablets BP 50mg
- Warfarin Tablets
- Nystatin Tablets
- Prednisolone Tablets
- Nystatin Vaginal Inserts
- Calcium Folinato
- Simvastatin Tablets
- Ferrous Sulphate And Folic Acid Tablet
- Colloidal bismuth subcitrate 120 mg tablets
- Colchicine Tablets
- Primaquine Tablets
- Amodiaquine Tablets
- Aceclofenac & Diacerein Tablets
- Artemether & Lumefantrine Tab
- VITAMIN B COMPLEX TABLETS
- Clotrimazole Tablet
- Tinidazole Tablets
- Fluconazole Tab
- ENALAPRIL MALEATE TABLETS USP 5MG
- Misoprostol Tablets 200 mcg
- Doxycyclin Hyclate Tablets USP 100 mg
- Dapsone Tablets BP 50 Mg
- Albendazole Tablets 400 Mg
- Calcium With Vitamin D3 Chewable Tablets
- Fluoxetine Tablet 20mg
- Erythromycin Stearate Tablets BP
- Aminophylline 100mg Tablets
- Lisinopril Hydrochlorothiazide tablets USP
- Ibuprofen Tablets 400mg
- Sulfadoxine Pyrimetanine Tablet
- Cefixime Tablets USP 200 mg
- Carvedilol Tablets
- Cefixime Tablets USP 400 mg
- Cefalexin Capsules BP 250mg
- Ciprofloxacin Tablets USP 750 mg
- Bisacodyl Tablets
- Prednisolone Tablets USP 20 mg
- Amitriptyline Tablets
- Nitrofurantoin Tablets BP
- Amoxicillin Dispersible Tablets
- Chloroquine Phosphate Tablets BP
- Ciprofloxacin Tablets USP 500
- Ciprofloxacin Tablets USP 500mg
- OLANZAPINE 10mg
- Prolonged - Release Potassium Chloride Tablets BP
- ALBENDAZOLE TABLETS USP 200mg
- Senna Tablets
- Ciprofloxacin Tablets USP 500 mg
- Gastro-resistant Diclofenac Tablets 50mg
- ClOPIDOGREL TABLETS USP 75mg
- Clopidogrel Tablets USP
- ALUMINIUM HYDROXIDE TABLETS BP 500mg
- Metronidazole Tablets BP 500mg
- Ranitidine Tablets USP
- Lisinopril Tablets USP 10mg
- Paracetamol Phenylephrine Hydrochloride and Chlorpheniramine Maleate Tablets
- Pyrimethamine Tablet
- Albendazole Tablets
- Carvedilol Tablets USP
- Carbimazole Tablets
- Lisinopril Tablets
- Griseofulvin Tablets BP
- Prednisolone Tablets BP
- Phenytoin Tablets BP
- Norfloxacin Tablets BP
- Ranitidine Tablet
- Levofloxacin Tablets
- Esomeprazole Tablets
- Devazole Tablet
- Fluconazole Tablet
- Paracetamol Phenylephrine HCl and Chlorpheniramine Maleate Tablets
- CLOTRIMAZOLE VAGINAL PESSARY
- Nifedipine Extended Release Tablets
- Betahistine Dihydrochloride Tablets
- Chloroquine Phosphate
- Azithromycin Tablets
- Dapsone Tablets
- sofosbuvir tablets
- Rivadev Tablet
- Devkabir Tablet
- Koldavin T
- Chloroquine Phosphate Tablets
- Pantoprazole Gastro-resistant Tablets BP 40 mg
- Sevlamer Hydrochloride Tablets
- Alfuzosin Prolonged Release Tablets
- Methyldopa Tablets BP
- Zinc Sulfate Tablets USP 20 mg
- Epiryte tablets
- Praziquantel Tablets USP 600 mg
- Salbutamol Tablets BP 2 mg
- Daclatasvir 60 mg & Sofosbuvir 400 mg Tablets
- Enlekavir Tablets USP 0.5 mg
- Clomifene Tablets BP 50 mg
- Leflunomide Tablets BP 10 mg
- Epiryte 500
- Pantoprazole Gastroresistant tablets
- Pentoxifylline Tablets
- Chloroquine Phosphate Tablets
- IMOD EV
- Azithromycin Tablets USP
- Charcoal Tablets 250 mg
- Carbimazole Tablets
- Amlodipine Besilate Tablets BP 5 mg
- Pharmaceutical Injections
- Chlorpromazine Injection
- Dobutamine Injection
- Lidocaine Injection
- Promethazine Injection
- Phenytoin Sodium Injections
- Kanamycin Injection
- Progesterone injection
- Prednisolone Acetate Injectable Suspension USP
- Sterilised Water for Injection
- Iron Sucrose Injection
- Bupivacaine Hcl with Dextrose Injection USP
- DEVCEF 1000 (Ceftriaxone for Injection USP 1000 mg)
- Cloxacillin Injection
- Adrenaline Injection
- Atenolol Injection
- Bupivacaine Injection
- Calcium Gluconate Injection
- Ceftriaxone Injection
- Magnesium Sulfate Injection USP 200mg
- Andrenaline Inj. BP
- Potassium Chloride for Injection Concentrate USP 100 mg/mL
- HEPARIN SODIUM INJECTION BP 5000 UI/ML
- Dopamine Hydrochloride Injection USP 200mg
- Clindamycin Injection
- CLINDAMYCIN INJECTION USP 150mg/ml
- Gentamycin Sulphate Injection
- Medroxyprogesterone Acetate Injection
- Dobutamine Injection USP 12.5 mg/mL
- Iron Dextran Injections
- Devoxa Injection
- Bupivacaine hcl in dextrose injection USP
- Vancomycin Hcl Injection
- Pantoprazole For Injection 40mg
- Imipramine HCL Injection
- Ceftriaxone For Injection USP 1g
- Suxamethonium Chloride Injection BP 50 mg/mL
- Vondan Ondansetron Injection
- Vancomycin Hydrochloride For Injection
- Adrenaline Injection BP 1mg
- Magnesium Sulfate Injection USP 500mg
- Hydralazine HCL Injection
- Noradrenaline Injection
- Potassium Chloride Injection
- Clonidine HCL Injection
- Amphotericin B
- Methylcobalamine Injection
- Sterile Potassium Chloride Concentrate
- Phenytoin Sodium Injection
- Ranitidine Injections
- Furosemide Injection
- Ondansetron Injection
- Ranitidine Injection
- Creams and Ointments
- Metronidazole with Povidone Iodine Cream
- Diclofenac Sodium Gel
- Betamethasone Valerate + Gentamicin Sulphate+Tonaftate Iodochlorhydroxyquinoline Cream
- Povidone Iodine Ointment U.S.P
- Lidocaine Jelly
- Crotamiton Cream
- Miconazole cream BP
- Hydroquinone Cream
- Silver Sulphadiazine Cream
- Tretinoin Cream
- Clobetasole Propionate Cream
- Fusidic Acid
- Acyclovir Ointment
- Capsaicin Gel
- Betamethasone Dipropionate Clotrimazole Neomycin Cream
- Anti-Hemorrhoidal Ointment 10 g
- Anti-Hemorrhoidal Ointment 20 g
- Zinc Oxide and Castor Oil Cream
- Beclomethasone Clotrimazole Neomycin Cream
- Beclomethasone / Clotrimazole / Neomycin Cream
- Capsin Gel
- Khwaderm Cream
- Miconazole Nitrate Cream
- Acyclovir Ointments
- 4 Ache
- Alpha Linolenic Acid, Diclofenac Diethylamine, Methyl Salicylate & Menthol Gel
- Hard Gelatin Capsules
- Amoxicillin Capsules BP 500mg
- FLUCLOXACLLIN CAPSULES BP 500mg
- Cefalexin Capsules BP 500mg
- Cefuroxime Axetile Tablets USP 250mg
- Fluconazole Capsules 200 mg
- Doxycycline Capsules BP 100mg
- Esomeprazole 20mg
- Esomeprazole 40mg
- Fluconazole Capsules
- Methylcobalamin and Pregabalin Capsules
- Azithromycin Capsules
- Oral Suspension
- Azithromycin for oral suspension USP
- Cefixime for Oral Suspension USP 100 mg/5 mL
- Artemether and Lumefantrine Oral Suspension
- Ciprofloxacin Suspension
- Ibuprofen and Paracetamol Suspension
- Carbocisteine Suspension
- Peadiatric Multivitamin Drops
- Artemether and Lumefantrine For Oral Suspension
- Amoxicillin Oral Suspension BP 250 mg
- Fluconazole Oral Suspension 50mg/5ml
- Azithromycin For Oral Suspension USP
- Artemether & Lumefantrine 180/1080
- Cefixime For Oral Suspension USP
- Metronidazole Oral suspension BP 125mg/5ml
- Bromhexine Hydrochloride
- Cloxacillin Sodium
- Quinine Sulfate
- Bromhexine Hydrochloride Elixir
- Dr. Mol Paracetamol Oral Suspension
- Devalzyn Suspension
- Amoxicillin Oral Suspension BP
- Nystantin Oral Suspension
- Devnyst Suspension
- Lactulose Solution
- Liquid Orals
- Soft Gelatin Capsules
- Injection Large Volume Parenterals
- Pharmaceutical Capsules
- Dry Syrup
- Eye Drops / Ear Drops
- Antiseptic & Antiparasitic Lotions
- Pharmaceutical Tablets
- More Info
- Contact Us
Phenytoin Sodium Injection
MOQ : 50000 Boxes
Phenytoin Sodium Injection Specification
- Pacakaging (Quantity Per Box)
- 10x2 ml
- Origin of Medicine
- India
- Salt Composition
- Phenytoin Sodium
- Drug Type
- Injection
- Physical Form
- Liquid
- Dosage
- As per suggestion
- Suitable For
- Children
- Storage Instructions
- Cool & Dry Place
Phenytoin Sodium Injection Trade Information
- Minimum Order Quantity
- 50000 Boxes
- Payment Terms
- Cash on Delivery (COD), Cash Advance (CA), Letter of Credit at Sight (Sight L/C), Letter of Credit (L/C)
- Sample Policy
- Contact us for information regarding our sample policy
- Packaging Details
- 10 x 2ml Ampoules in printed carton with pack insert.
- Main Export Market(s)
- Asia, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Africa
- Main Domestic Market
- All India
About Phenytoin Sodium Injection
Indications:
Phenytoin Sodium Injection USP is indicated for the control of status epilepticus of the grand mal type, and prevention and treatment of seizures occurring during neurosurgery.
Dosage & Administration:
The addition of Phenytoin Sodium Injection USP to intravenous infusion is not recommended due to lack of solubility and resultant precipitation.
Not to exceed 50 mg per minute, intravenously in adults, and not exceeding 1 - 3 mg/kg/min in neonates.
Status Epilepticus
In adults, a loading dose of 10 to 15 mg/kg should be administered slowly intravenously, at a rate not exceeding 50 mg per minute. The loading dose should be followed by maintenance doses of 100 mg orally or intravenously every 6-8 hours.
Neurosurgery
Prophylactic dosage 100 to 200 mg intramuscularly at approximately 4-hour intervals during surgery and continued during the postoperative period.
Contraindications:
Phenytoin is contraindicated in patients with a history of hypersensitivity to hydantoin products. Because of its effect on ventricular automaticity, phenytoin is contraindicated in sinus bradycardia, sino-atrial block, second and third degree A-V block, and patients with Adams-Stokes syndrome.
Warnings:
Intravenous administration should not exceed 50 mg per minute in adults. In neonates, the drug should be administered at a rate not exceeding 1 - 3 mg/kg/min.
Severe cardiotoxic reactions and fatalities have been reported with atrial and ventricular conduction depression and ventricular fibrillation. Severe complications are most commonly encountered in elderly or gravely ill patients.
FAQs of Phenytoin Sodium Injection:
Q: What is the physical form of Phenytoin Sodium Injection?
A: The physical form of Phenytoin Sodium Injection is liquid.Q: How should Phenytoin Sodium Injection be stored?
A: Phenytoin Sodium Injection should be stored in a cool and dry place.Q: What is the salt composition of Phenytoin Sodium Injection?
A: The salt composition of Phenytoin Sodium Injection is Phenytoin Sodium.Q: Who is Phenytoin Sodium Injection suitable for?
A: Phenytoin Sodium Injection is suitable for children.Q: What is the packaging quantity per box of Phenytoin Sodium Injection?
A: The packaging quantity per box of Phenytoin Sodium Injection is 10x2 ml.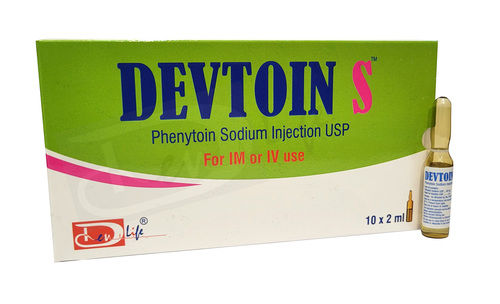
Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
More Products in Pharmaceutical Injections Category
Methylcobalamine Injection
Minimum Order Quantity : 80000 Pieces
Origin of Medicine : India
Physical Form : Liquid
Drug Type : Injection
Storage Instructions : Cool & Dry Place
Dosage : As per suggestion
Ranitidine Injections
Minimum Order Quantity : 50000 Boxes
Origin of Medicine : India
Physical Form : Liquid
Drug Type : Injection
Storage Instructions : Cool & Dry Place
Dosage : As per suggestion
Iron Sucrose Injection
Minimum Order Quantity : 10 Pieces
Origin of Medicine : India
Physical Form : Liquid
Drug Type : Injection
Storage Instructions : Cool & Dry Place
Dosage : As per suggestion
Cloxacillin Injection
Origin of Medicine : Synthetic
Physical Form : Other, Injection
Drug Type : Other, Antibiotic
Storage Instructions : Store in a cool dry place below 25C away from light and moisture
Dosage : To be administered as directed by a healthcare professional

 Send Inquiry
Send Inquiry

 Call Me Free
Call Me Free
