- Home Page
- Company Profile
-
Our Products
- Pharmaceutical Tablets
- Sulfadiazine Tablet
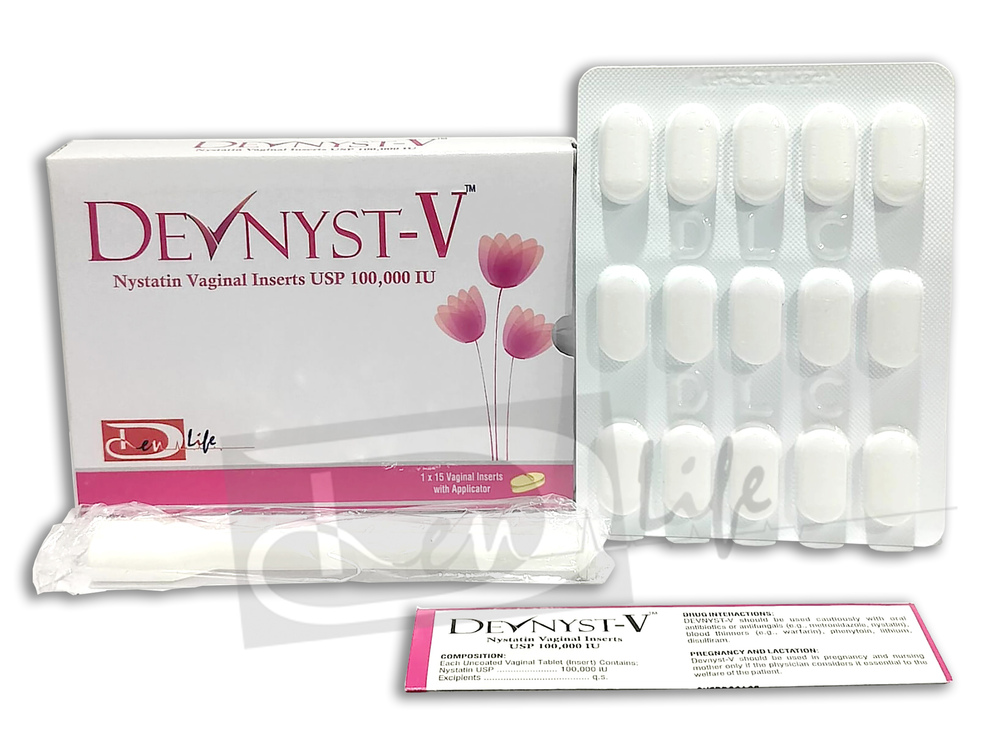
- Nystatin Vaginal Inserts
- Simvastatin Tablets
- Clotrimazole Tablet
- Clomifene Tablets BP 50mg
- Ciprofloxacin Tablets 500mg
- Ferrous Sulphate And Folic Acid Tablet
- Nystatin Tablets
- Sodium Bicarbonate Tablets
- Colloidal bismuth subcitrate 120 mg tablets
- Primaquine Tablets
- Cefuroxime Axetile Tablets
- Prednisolone Tablets
- Vitamin D3 Tablets
- Prochlorperazine Tablets
- Ferrous Sulphate and Folic Acid
- Amodiaquine Tablets
- Colchicine Tablets
- Aceclofenac & Diacerein Tablets
- Artemether & Lumefantrine Tab
- Fexofenadine HCL Tablets
- Fluconazole Tab
- Tinidazole Tablets
- Calcium Folinato
- Warfarin Tablets
- Quinine Sulphate Tablet 300 mg
- Nifedipine Extended Release Tablets USP 20 mg
- Artemether and Lumefantrine Tablets
- VITAMIN B COMPLEX TABLETS
- ENALAPRIL MALEATE TABLETS USP 5MG
- MIstop Talets
- Doxycyclin Hyclate Tablets USP 100 mg
- Calcium With Vitamin D3 Chewable Tablets
- Dapsone Tablets BP 50 Mg
- Albendazole Tablets 400 Mg
- Aminophylline 100mg Tablets
- Fluoxetine Tablet 20mg
- Erythromycin Stearate Tablets BP
- Lisinopril Hydrochlorothiazide tablets USP
- Cefixime Tablets USP 200 mg
- Ibuprofen Tablets 400mg
- Sulfadoxine Pyrimetanine Tablet
- Cefixime Tablets USP 400 mg
- Carvedilol Tablets
- Cefalexin Capsules BP 250mg
- Ciprofloxacin Tablets USP 750 mg
- Prednisolone Tablets USP 20 mg
- Bisacodyl Tablets
- Amitriptyline Tablets
- Nitrofurantoin Tablets BP
- Ciprofloxacin Tablets USP 500
- Chloroquine Phosphate Tablets BP
- Amoxicillin Dispersible Tablets
- Prolonged - Release Potassium Chloride Tablets BP
- Ciprofloxacin Tablets USP 500mg
- ALBENDAZOLE TABLETS USP 200mg
- OLANZAPINE 10mg
- Gastro-resistant Diclofenac Tablets 50mg
- Senna Tablets
- Ciprofloxacin Tablets USP 500 mg
- Clopidogrel Tablets USP
- ClOPIDOGREL TABLETS USP 75mg
- ALUMINIUM HYDROXIDE TABLETS BP 500mg
- Metronidazole Tablets BP 500mg
- Ranitidine Tablets USP
- Lisinopril Tablets USP 10mg
- Paracetamol Phenylephrine Hydrochloride and Chlorpheniramine Maleate Tablets
- Pyrimethamine Tablet
- Albendazole Tablets
- Carvedilol Tablets USP
- Carbimazole Tablets
- Lisinopril Tablets
- Prednisolone Tablets BP
- Griseofulvin Tablets BP
- Phenytoin Tablets BP
- Norfloxacin Tablets BP
- Ranitidine Tablet
- Levofloxacin Tablets
- Esomeprazole Tablets
- Devazole Tablet
- Fluconazole Tablet
- Paracetamol Phenylephrine HCl and Chlorpheniramine Maleate Tablets
- CLOTRIMAZOLE VAGINAL PESSARY
- Nifedipine Extended Release Tablets
- Betahistine Dihydrochloride Tablets
- Chloroquine Phosphate
- Azithromycin Tablets
- Dapsone Tablets
- sofosbuvir tablets
- Rivadev Tablet
- Devkabir Tablet
- Koldavin T
- Chloroquine Phosphate Tablets
- Devpantop
- Sevlamer HYdrochloride Tablets
- Alfuzosin Prolonged Release Tablets
- Methyldopa Tablets BP
- Zinc Sulfate Tablets USP 20 mg
- Gastro Resistant Sodium Valproate Tablets BP 200mg
- Praziquantel Tablets USP 600 mg
- Salbutamol Tablets BP 2 mg
- Daclatasvir 60 mg & Sofosbuvir 400 mg Tablets
- Enlekavir Tablets USP 0.5 mg
- Clomifene Tablets BP 50 mg
- Leflunomide Tablets BP 10 mg
- Epiryte 500
- Pantoprazole Gastroresistant tablets
- Chloroquine Phosphate Tablets
- Pharmaceutical Injections
- Chlorpromazine Injection
- Dobutamine Injection
- Lidocaine Injection
- Promethazine Injection
- Phenytoin Sodium Injections
- Kanamycin Injection
- Progesterone injection
- Prednisolone Acetate Injectable Suspension USP
- Sterilised Water for Injection
- Iron Sucrose Injection
- Bupivacaine Hcl with Dextrose Injection USP
- DEVCEF 1000 (Ceftriaxone for Injection USP 1000 mg)
- Cloxacillin Injection
- Adrenaline Injection
- Atenolol Injection
- Bupivacaine Injection
- Calcium Gluconate Injection
- Ceftriaxone Injection
- Magnesium Sulfate Injection USP 200mg
- Andrenaline Inj. BP
- Potassium Chloride for Injection Concentrate USP 100 mg/mL
- HEPARIN SODIUM INJECTION BP 5000 UI/ML
- Dopamine Hydrochloride Injection USP 200mg
- Clindamycin Injection
- CLINDAMYCIN INJECTION USP 150mg/ml
- Gentamycin Sulphate Injection
- Medroxyprogesterone Acetate Injection
- Dobutamine Injection USP 12.5 mg/mL
- Iron Dextran Injections
- Devoxa Injection
- Bupivacaine hcl in dextrose injection USP
- Vancomycin Hcl Injection
- Pantoprazole For Injection 40mg
- Imipramine HCL Injection
- Ceftriaxone For Injection USP 1g
- Suxamethonium Chloride Injection BP 50 mg/mL
- Vondan Ondansetron Injection
- Vancomycin Hydrochloride For Injection
- Adrenaline Injection BP 1mg
- Magnesium Sulfate Injection USP 500mg
- Hydralazine HCL Injection
- Noradrenaline Injection
- Potassium Chloride Injection
- Clonidine HCL Injection
- Amphotericin B
- Methylcobalamine Injection
- Sterile Potassium Chloride Concentrate
- Phenytoin Sodium Injection
- Ranitidine Injections
- Furosemide Injection
- Ondansetron Injection
- Ranitidine Injection
- Creams and Ointments
- Diclofenac Sodium Gel
- Betamethasone Valerate + Gentamicin Sulphate+Tonaftate Iodochlorhydroxyquinoline Cream
- Povidone Iodine Ointment U.S.P
- Metronidazole with Povidone Iodine Cream
- Lidocaine Jelly
- Crotamiton Cream
- Miconazole cream BP
- Hydroquinone Cream
- Silver Sulphadiazine Cream
- Tretinoin Cream
- Clobetasole Propionate Cream
- Fusidic Acid
- Acyclovir Ointment
- Capsaicin Gel
- Betamethasone Dipropionate Clotrimazole Neomycin Cream
- Anti-Hemorrhoidal Ointment 10 g
- Anti-Hemorrhoidal Ointment 20 g
- Zinc Oxide and Castor Oil Cream
- Beclomethasone Clotrimazole Neomycin Cream
- Beclomethasone / Clotrimazole / Neomycin Cream
- Capsin Gel
- Khwaderm Cream
- Miconazole Nitrate Cream
- Acyclovir Ointments
- Hard Gelatin Capsules
- Amoxicillin Capsules BP 500mg
- FLUCLOXACLLIN CAPSULES BP 500mg
- Cefalexin Capsules BP 500mg
- Cefuroxime Axetile Tablets USP 250mg
- Fluconazole Capsules 200 mg
- Doxycycline Capsules BP 100mg
- Esomeprazole 20mg
- Esomeprazole 40mg
- Fluconazole Capsules
- Methylcobalamin and Pregabalin Capsules
- Azithromycin Capsules
- Oral Suspension
- Ibuprofen and Paracetamol Suspension
- Azithromycin for oral suspension USP
- Cefixime for Oral Suspension USP 100 mg/5 mL
- Artemether and Lumefantrine Oral Suspension
- Ciprofloxacin Suspension
- Carbocisteine Suspension
- Peadiatric Multivitamin Drops
- Artemether and Lumefantrine For Oral Suspension
- Amoxicillin Oral Suspension BP 250 mg
- Fluconazole Oral Suspension 50mg/5ml
- Artemether & Lumefantrine 180/1080
- Azithromycin For Oral Suspension USP
- Cefixime For Oral Suspension USP
- Metronidazole Oral suspension BP 125mg/5ml
- Bromhexine Hydrochloride
- Cloxacillin Sodium
- Quinine Sulfate
- Bromhexine Hydrochloride Elixir
- Dr. Mol Paracetamol Oral Suspension
- Devalzyn Suspension
- Liquid Orals
- Soft Gelatin Capsules
- Injection Large Volume Parenterals
- Pharmaceutical Capsules
- Dry Syrup
- Eye Drops / Ear Drops
- Antiseptic & Antiparasitic Lotions
- Pharmaceutical Tablets
- More Info
- Contact Us
Lisinopril Hydrochlorothiazide tablets USP
MOQ : 10 Pieces
Lisinopril Hydrochlorothiazide tablets USP Specification
- Pacakaging (Quantity Per Box)
- 10x10 Tablets
- Origin of Medicine
- India
- Salt Composition
- Lisinopril, Hydrochlorothiazide
- Drug Type
- General Medicines
- Ingredients
- Each uncoated tablet contains: Lisinopril USP..............................10 mg Hydrochlorothiazide USP.......12.5 mg Excipients..........................................q.s.
- Physical Form
- Tablets
- Dosage
- As directed by Physician
- Suitable For
- Adults
- Storage Instructions
- Store in a cool and dry place away from light
Lisinopril Hydrochlorothiazide tablets USP Trade Information
- Minimum Order Quantity
- 10 Pieces
- Supply Ability
- 100 Pieces Per Month
- Delivery Time
- 7 Days
- Packaging Details
- 10 X 10 BLISTER IN CARTON WITH PACK INSERT
About Lisinopril Hydrochlorothiazide tablets USP
DEVAPRIL-H
Lisinopril Hydrochlorothiazide tablets USP
COMPOSITION:
Eachuncoated tablet contains:- Lisinopril USP............................10 mg
- Hydrochlorothiazide USP........12.5 mg
- Excipients.......................................q.s.
INDICATION:
Lisinopril / hydrochlorothiazide is indicated in the management of mild to moderate hypertension in patients who have been stabilised on the individual components given in the same proportions.
PRESENTATION:
10 x 10 tablets in printed carton with pack insert.
Product details
|
Shelf life |
36 MONTHS |
|
Form |
Tablet |
|
Manufacturer |
DEV LIFE CORPORATION |
|
Brand |
DEVAPRIL-H |
|
Pack size |
10 X 10 |
|
Packaging Type |
10 X 10 BLISTER |
|
Composition |
COMPOSITION Each uncoated tablet contains Lisinopril USP eq. to Lisinopril Anhydrous...10mg Ex |
FAQs of Lisinopril Hydrochlorothiazide tablets USP:
Q: What is the recommended dosage for Lisinopril Hydrochlorothiazide tablets USP?
A: The dosage should be taken as directed by a physician.Q: Where should Lisinopril Hydrochlorothiazide tablets USP be stored?
A: The tablets should be stored in a cool and dry place away from light.Q: What is the origin of Lisinopril Hydrochlorothiazide tablets USP?
A: The medicine originates from India.Q: How many tablets are there in each box of Lisinopril Hydrochlorothiazide tablets USP?
A: Each box contains 10x10 tablets.Q: What is the salt composition of Lisinopril Hydrochlorothiazide tablets USP?
A: The tablets contain Lisinopril and Hydrochlorothiazide as the salt composition.
Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
More Products in Pharmaceutical Tablets Category
Nystatin Vaginal Inserts
Minimum Order Quantity : 1 Box
Suitable For : Adults
Physical Form : Other, Vaginal Insert (suppository/tablet)
Origin of Medicine : Allopathic
Storage Instructions : Store below 25C, protect from moisture and direct sunlight
Salt Composition : Nystatin
Prednisolone Tablets BP
Suitable For : Other, Adults and children (as prescribed).
Physical Form : Tablets
Origin of Medicine : Synthetic
Storage Instructions : Store in a cool dry place below 25C. Protect from light and moisture.
Salt Composition : Prednisolone BP
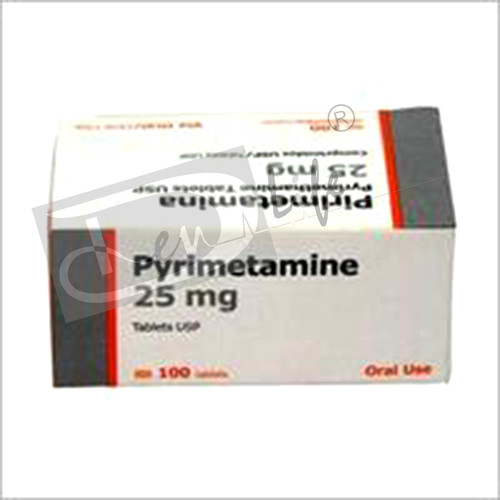
Pyrimethamine Tablet
Suitable For : Other, Adults and children as prescribed by a healthcare professional
Physical Form : Other, Tablet
Origin of Medicine : Allopathic
Storage Instructions : Store in a cool dry place away from direct sunlight
Salt Composition : Pyrimethamine
Nitrofurantoin Tablets BP
Suitable For : Other, Adults and children as recommended by the physician
Physical Form : Other, Tablet
Origin of Medicine : Synthetic
Storage Instructions : Store at room temperature (15C to 30C) protect from moisture and light
Salt Composition : Nitrofurantoin Sodium / Nitrofurantoin Monohydrate

 Send Inquiry
Send Inquiry


 Call Me Free
Call Me Free
